

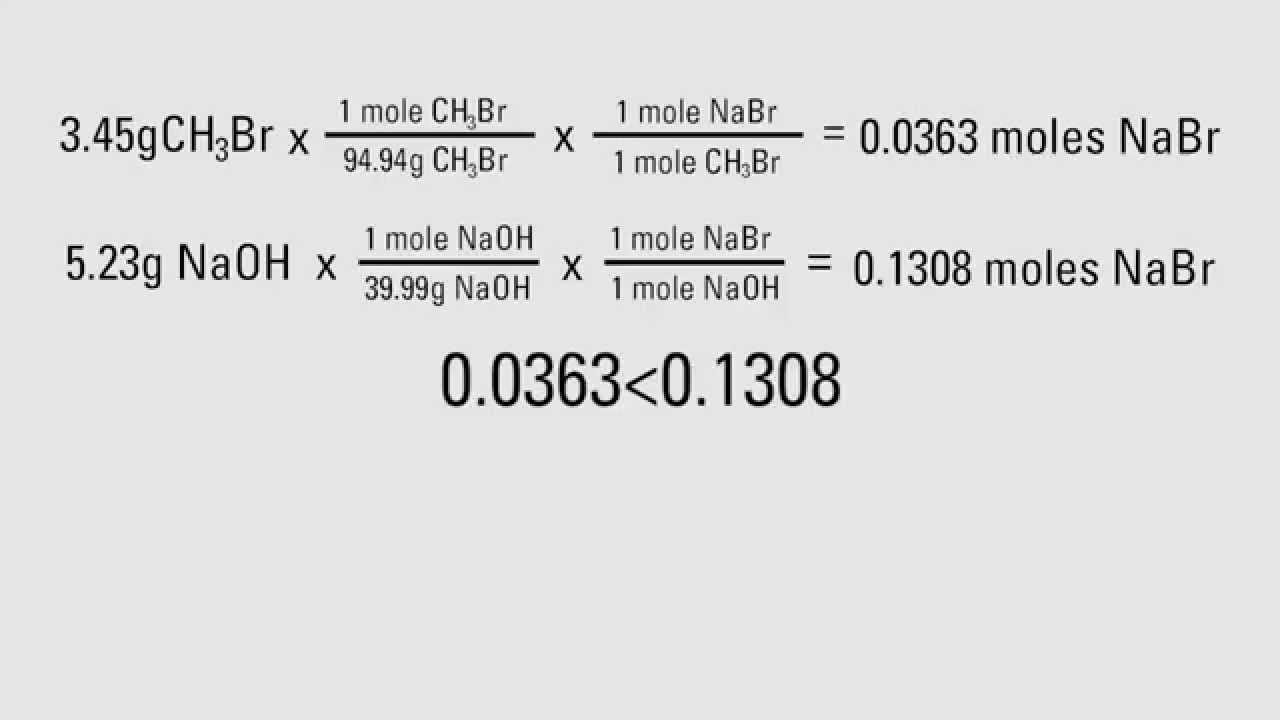
On the other hand, from the reaction equation, we see that for 1 molecule of CaCO 3 there are 2 molecules of HCl. Molecular weights of CaCO 3 and HCl are 100.0869 g/mol and 36.46094 g/mol respectively.
What is the theoretical yield of calcium chloride resulting from this chemical reaction?įirst write the following balanced chemical equation: Example of a Chemical ReactionĬonsider reaction of 10.5 g of calcium carbonate with 13.6 g of hydrochloric acid. Its amount determines the amount of reaction products, or the theoretical yield. The reagent in short supply will be the limiting reactant. Then the values found must be compared with the stoichiometric coefficients of the chemical reaction in question.īy comparing the proportions of these values it is possible to determine which reagents are in excess and which reagent is in deficit. To find the limiting reactant, it is necessary to find out how many moles of the initial reactants are involved in the reaction. The limiting reactant is the one that is in short supply, so that the reaction cannot proceed when it is used up. This is based on the molar ratio between the reagents. In order to calculate the theoretical yield, you must first find the limiting reactant in a balanced chemical equation. The theoretical yield is commonly expressed in grams or moles. In practice, it is usually different from the amount that you actually get from the reaction in the laboratory. This is the amount of product that can theoretically be obtained as a result of an ideal chemical reaction. The theoretical yield is the amount of product obtained from the complete consumption of the limiting reactant in a chemical reaction.

